About us
About Lindo Life Science
Lindo Life Science is a healthcare focused life science company that develops antimicrobial technologies to support infection prevention and improve patient outcomes in clinical environments. As part of the Lindo Technology Group, we combine scientific research, clinical insight and medical device development to address some of healthcare’s most persistent infection challenges.
Importantly, our work starts with a simple belief. Infection prevention should begin at the source. Therefore, we focus on preventative technologies that are effective, practical and designed for real world clinical use.

A focused life science practice
Lindo Life Science translates antimicrobial blue light science into practical medical technologies. Our team focuses on developing solutions that integrate safely into clinical workflows while meeting the rigorous standards required for healthcare use.
Our work spans early stage research, product development, usability engineering and regulatory planning. By working closely with clinicians, researchers and industry partners, we ensure our technologies address genuine clinical needs and real clinical constraints. Our focus is for our technology to fit the clinical process, not the other way around.
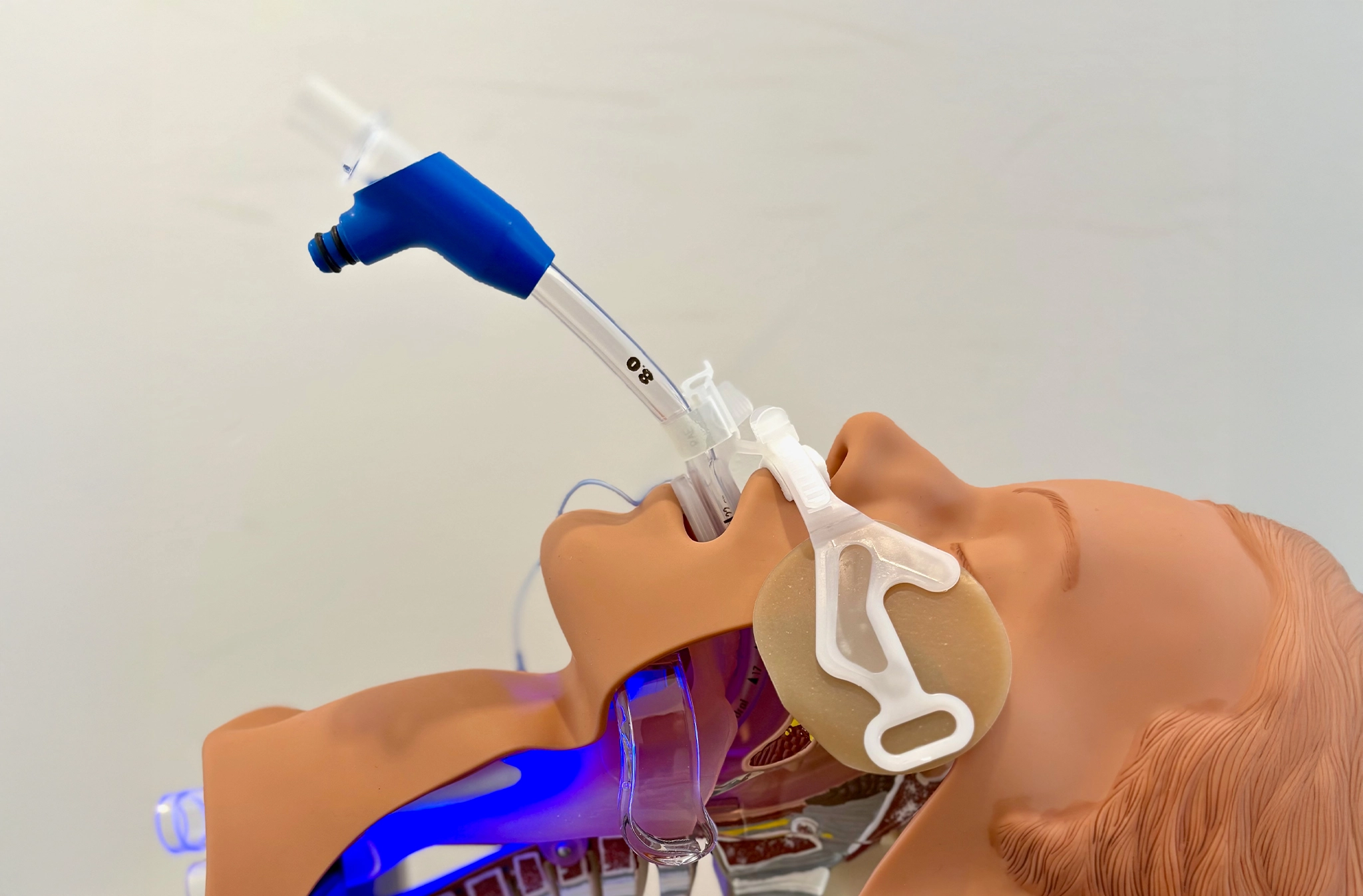
As a result, the Lindo Tube, our first medical device in development, targets bacterial contamination within ventilator circuits. Through this approach, the device aims to reduce the risk of ventilator associated pneumonia in critically ill patients.
Partnerships and progress
Lindo Life Science is advancing its medical device portfolio through a strong foundation of clinical collaboration, strategic partnerships and government support. The business is supported by matched AU$2 million funding from the Australian Government’s Industry Growth Program and operates under a strategic know how licence agreement with Mayo Clinic, enabling close collaboration with clinicians in the United States.
Our broader partner network includes leading organisations across medical device design, engineering, regulatory strategy and translational research. These partnerships allow us to move efficiently from scientific concept to clinically relevant solutions while maintaining a clear pathway toward regulatory approval and commercialisation.
Our work is delivered by a multidisciplinary team spanning science, engineering, clinical collaboration and regulatory expertise. Together, the team brings deep experience in developing healthcare technologies designed for real world clinical environments.

Our investment partners and project collaborators








The Lindo Group
Lindo Technology Group was founded with a clear purpose to apply light based technologies to improve health, safety and wellbeing across a variety of environments. As research into antimicrobial blue light advanced, the opportunity to apply this science within healthcare and medical devices became increasingly clear.
In response, the Lindo Life Science division was established as a dedicated practice focused exclusively on life sciences and healthcare applications. This has allowed the team to concentrate on regulated clinical environments, medical device development and partnerships with leading healthcare institutions.
Today, Lindo Life Science represents the fastest growing area of the Lindo Group and a core pillar of its future direction.
What sets us apart
Science led development
Our technologies are grounded in established antimicrobial blue light research and supported by ongoing collaboration with academic and clinical partners. We focus on mechanisms that reduce bacteria without contributing to antimicrobial resistance.
Clinical collaboration
We work directly with healthcare professionals to ensure our solutions are designed for real clinical environments. Partnerships with institutions such as Mayo Clinic inform our development process from concept through to clinical readiness.
Medical device focus
Lindo Life Science is structured around the requirements of medical device development, including usability, safety, quality systems and regulatory pathways. This ensures our technologies are developed with global healthcare markets in mind.
Non chemical infection prevention
Our approach avoids reliance on chemicals or antibiotics, offering an alternative pathway to bacterial reduction that supports broader antimicrobial stewardship goals.
Global perspective with Australian capability
While our technologies are designed for global healthcare systems, development and delivery are grounded in Australian capability, supporting sovereign manufacturing and advanced medical technology development.
Light with impact
Get in touch to see how we can collaborate on a bespoke Lindo solution to suit your needs.
